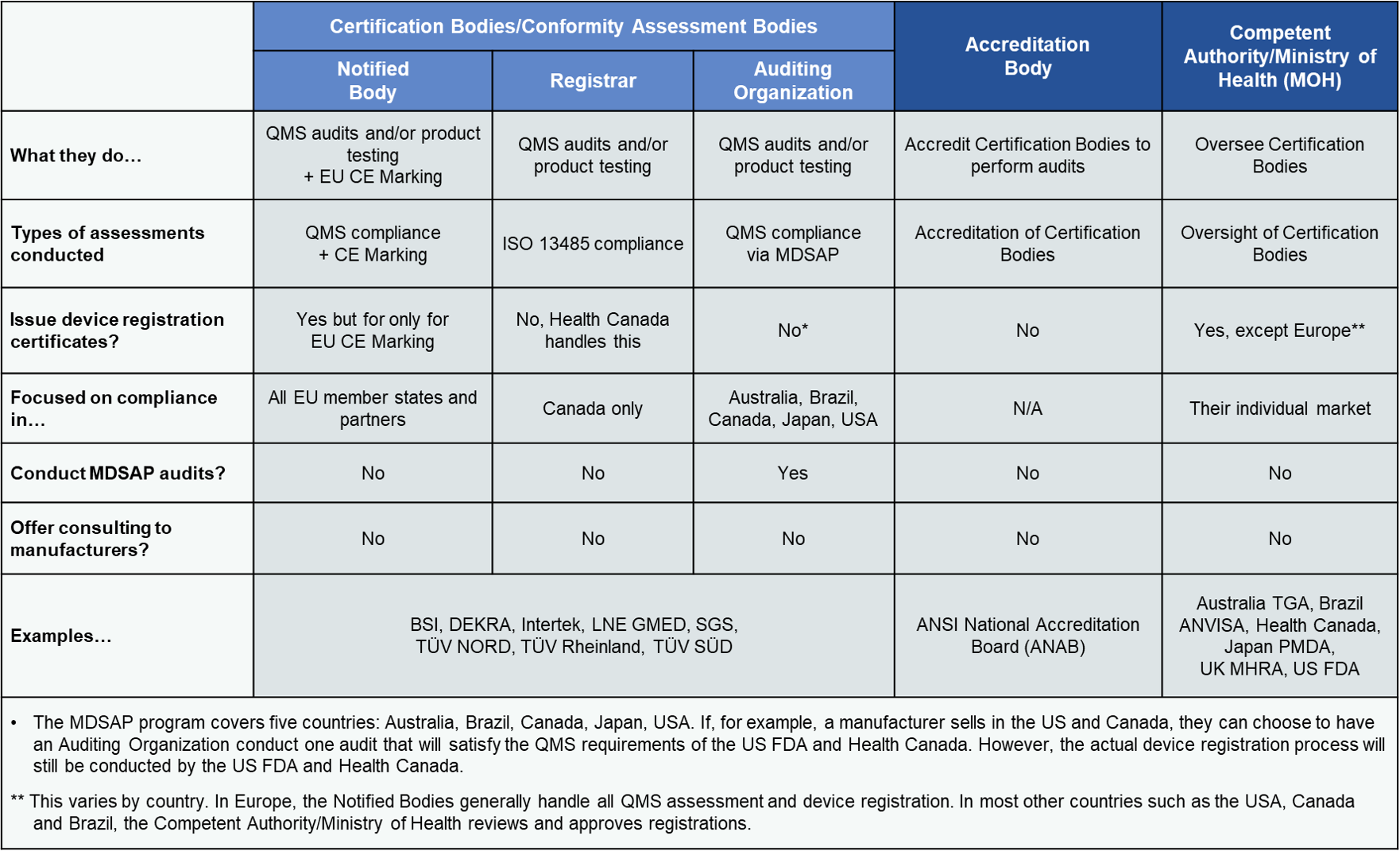
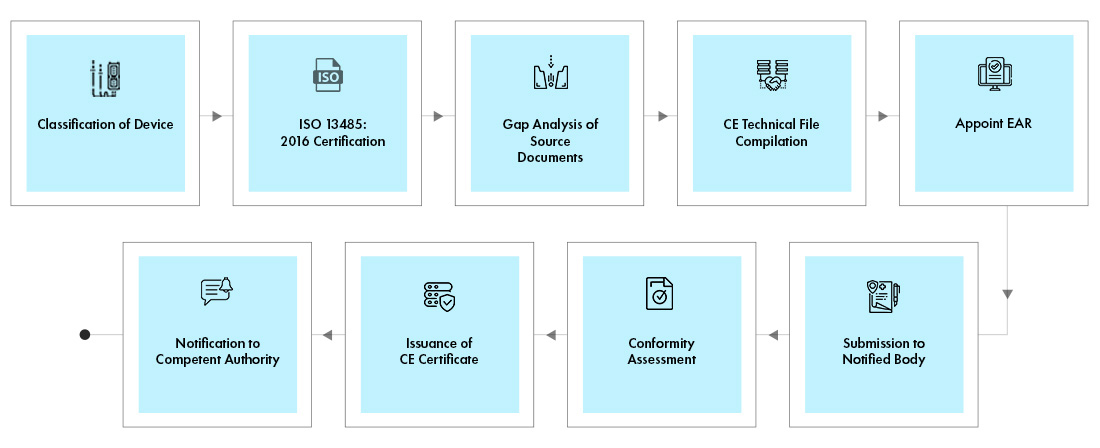
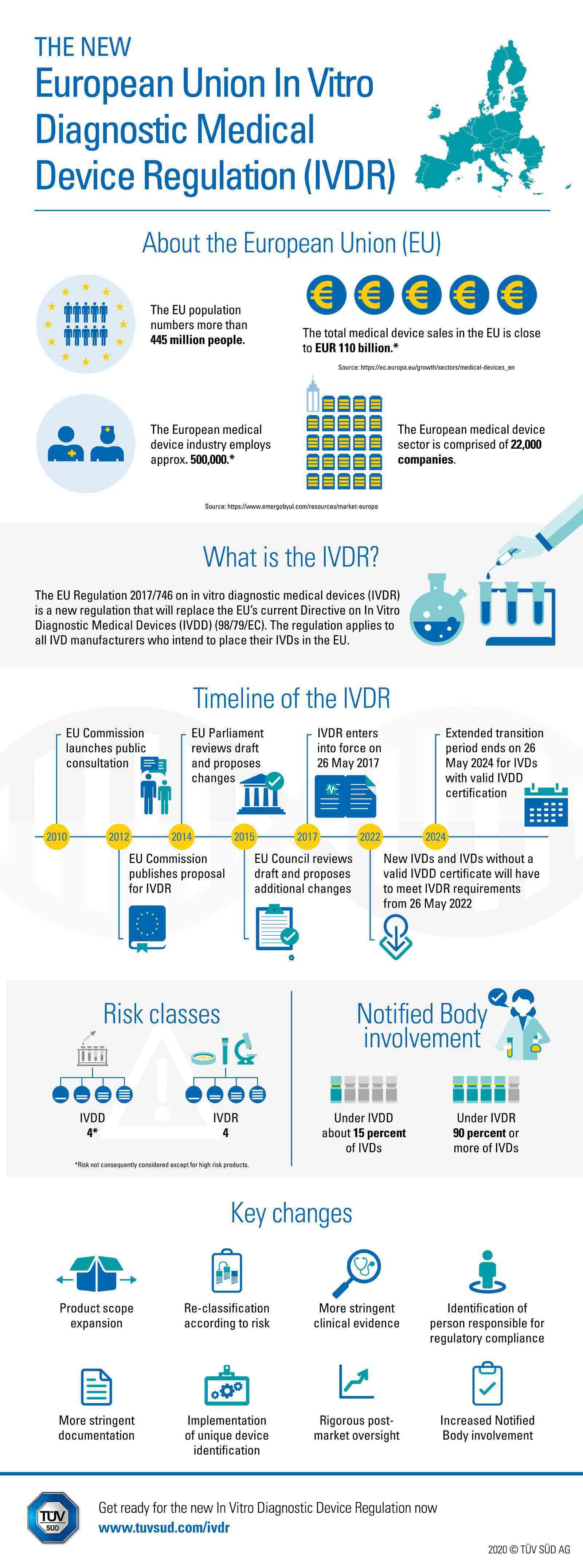
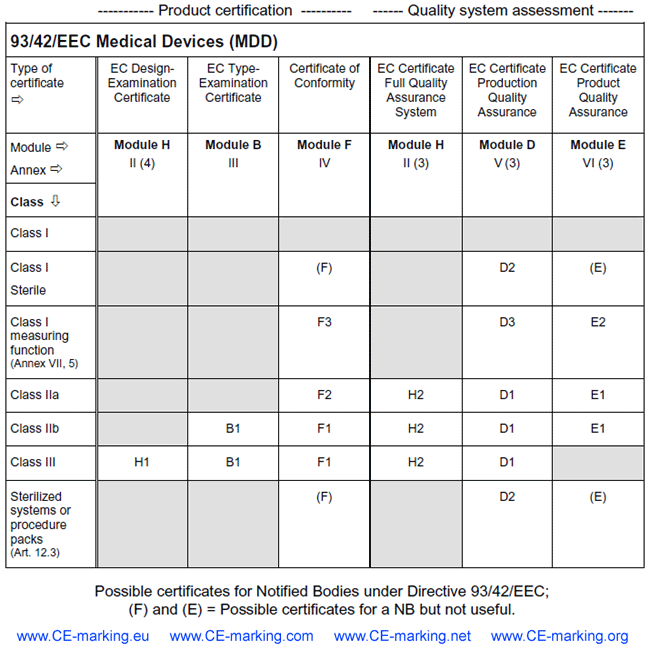
Competent Authority, Notified Body, ISO Registrar: How Each Role Functions in the Medical Device Industry

Safeguarding public health Implementing Medical Device Regulation and Maintaining Patient Safety Name Nicola Lennard Date October ppt download










![EU MDR Update: How To Get a New Medical Device Certified? [Flow Charts] - Sofeast EU MDR Update: How To Get a New Medical Device Certified? [Flow Charts] - Sofeast](https://www.sofeast.com/wp-content/uploads/2021/08/class-I-medical-devices.jpeg)
/tuv-rheinland-ivdr-visual-2-en_core_1_x.png)