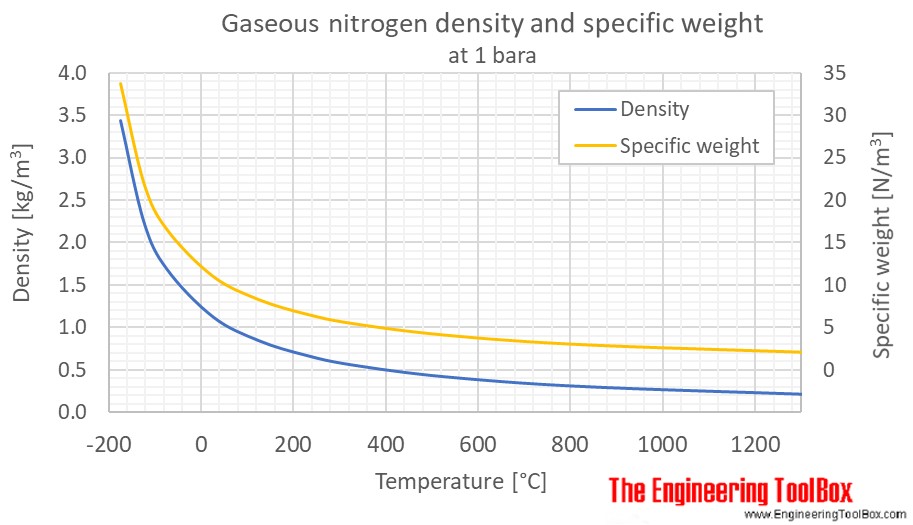
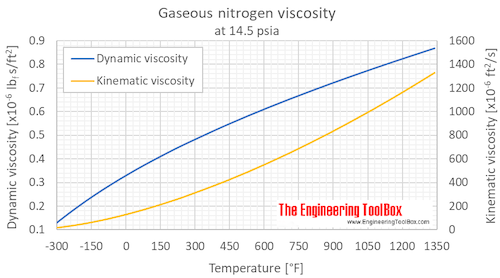
Nitrogen reacts with hydrogen to give ammonia. Calculate the volume of the ammonia gas formed when nitrogen reacts with 6 litres of hydrogen. All volumes measured at S.T.P.

Calculate the mass of `3.011 xx 10^(24)` molecules of nitrogen gas `(N_(2))`. (Atomic mass : `N = 14 - YouTube