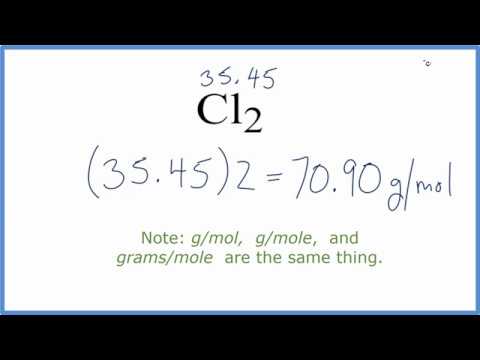Calculate the molecular mass of chloroform `(CHCl_(3))`. (Atomic masses : `C = 12 u , H = 1 u , Cl = - YouTube
Cl Chlorine Chemical Element Periodic Table. Single vector illustration, colorful Icon with molar mass, electron conf. and atomic number Stock Vector Image & Art - Alamy

Molar mass of 223.94 g/mol consists of 32.18% C, 4.50% H, and 63.32% Cl Find the molecular formula - Brainly.com

SOLVED: how to calculate the uncertainty of molar mass of Cl- and any other element in the periodic table??
✓ Solved: Papaverine hydrochloride (abbreviated papH^+Cl^-; molar mass=378.85g∕mol) is a drug that belongs...
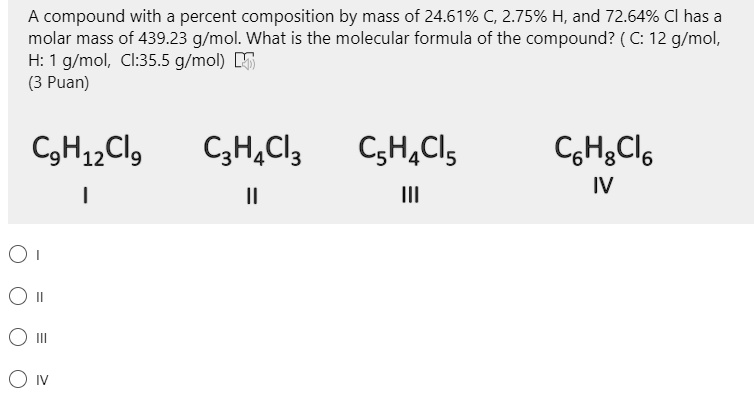






![Stoffmengekonzentration • Formel und Berechnung · [mit Video] Stoffmengekonzentration • Formel und Berechnung · [mit Video]](https://d1g9li960vagp7.cloudfront.net/wp-content/uploads/2022/05/Stoffmengenkonzentration--1024x576.jpg)