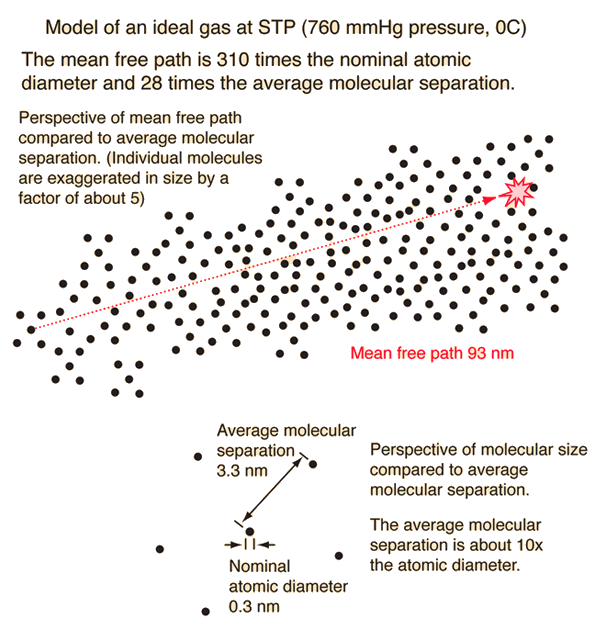
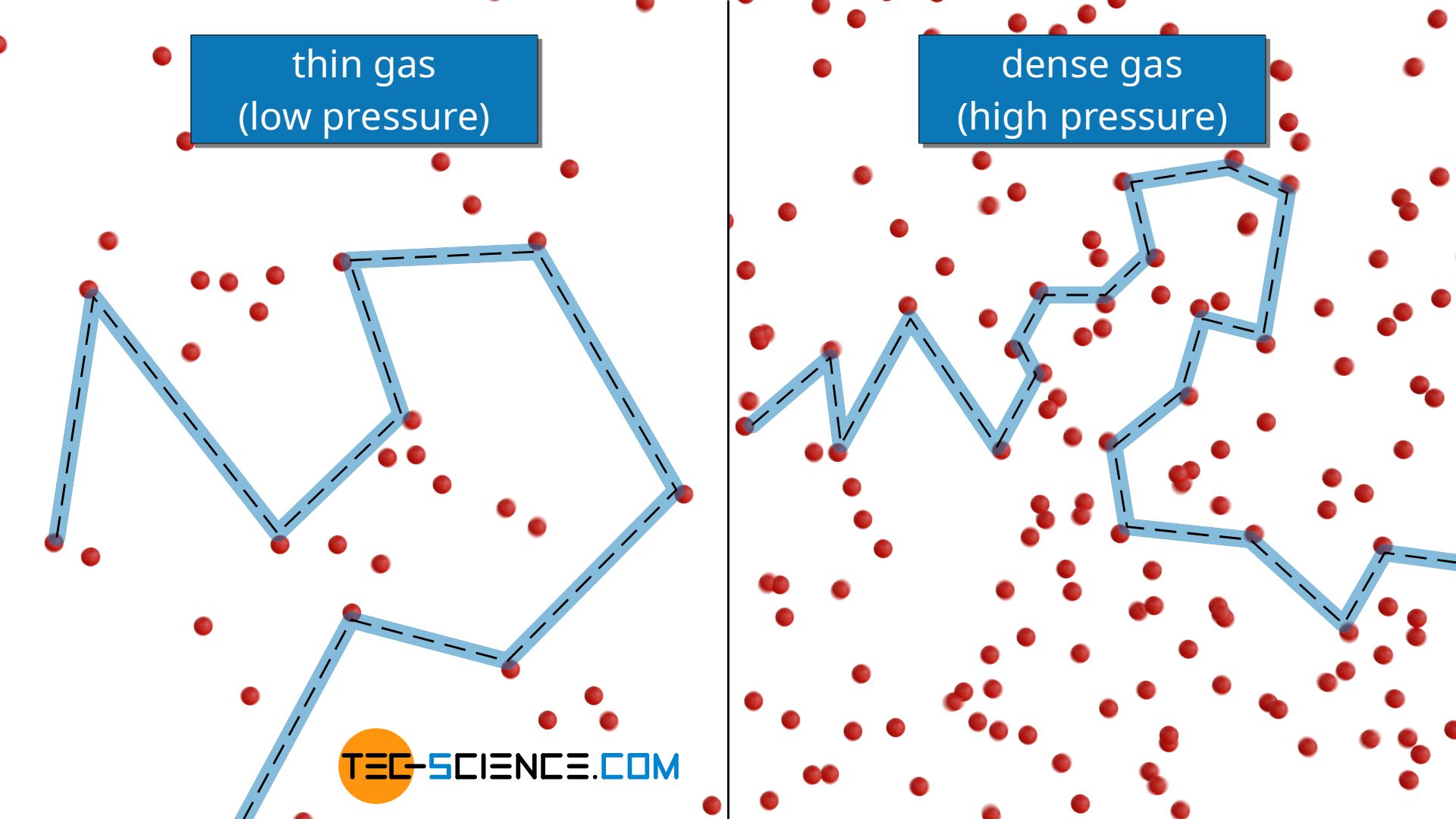
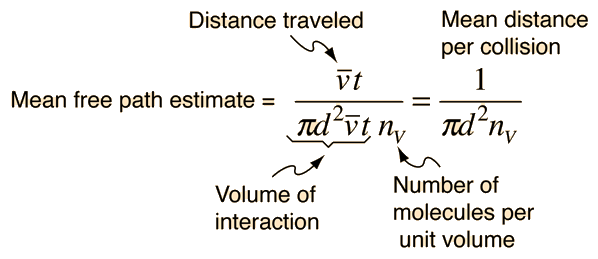
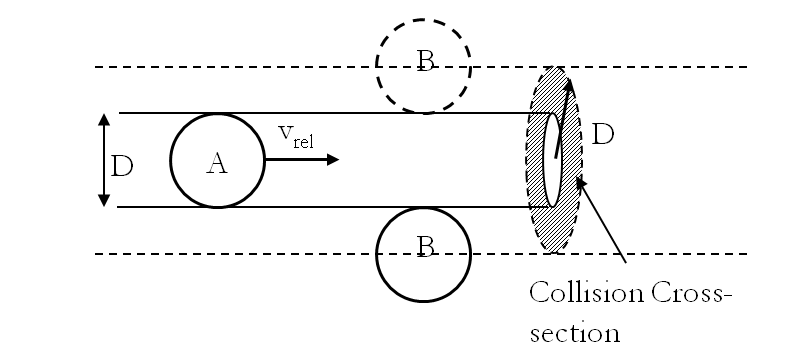
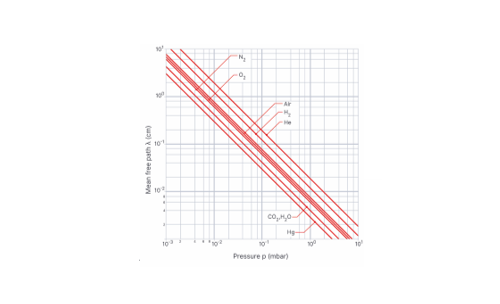
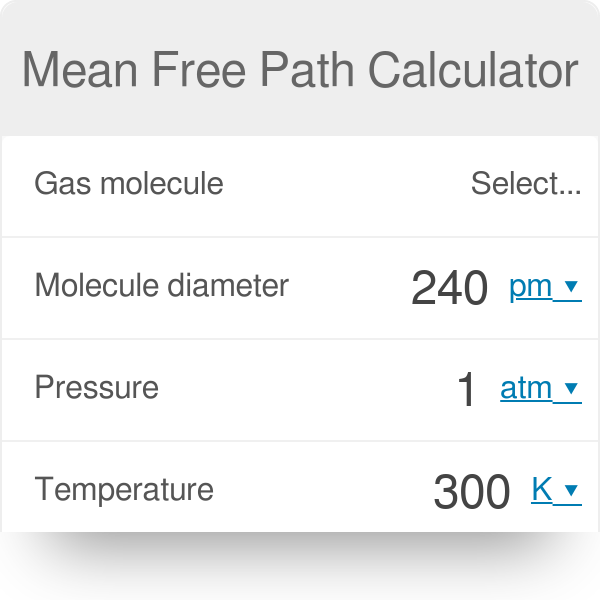
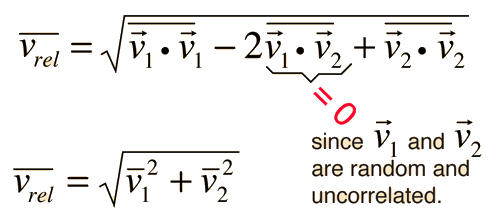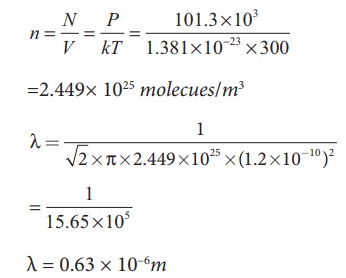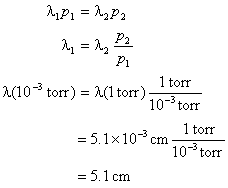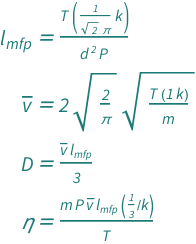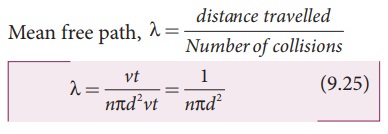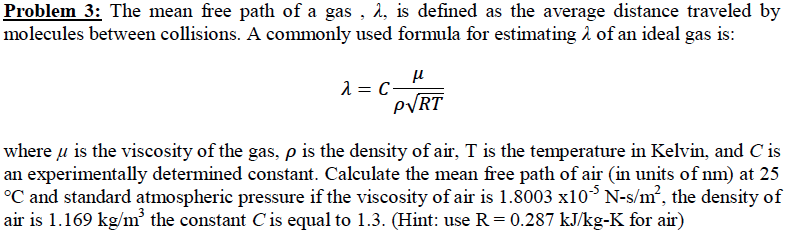The effect of system boundaries on the mean free path for confined gases: AIP Advances: Vol 3, No 10

Mean Free Path, Mean Free Time, & Root Mean Square Velocity Formula Chemistry & Physics Problems - YouTube

Calculate the means free path of nitrogen molecule at 27^o C when pressure is 1.0 atm. Given, diameter of nitrogen molecule = 1.5 oA , kB = 1.38 × 10^-23 J K^-1 .
![Calculate the mean free path in CO2 at 27^∘C and a pressure of 10^-9 bar. Molecular diameter of CO2 is 500 pm.[ Given : R = 25/3 J.mol^-1K^-1, √(2) = 1.4, pi = 22/7, NA = 6 × 10^23 ] Calculate the mean free path in CO2 at 27^∘C and a pressure of 10^-9 bar. Molecular diameter of CO2 is 500 pm.[ Given : R = 25/3 J.mol^-1K^-1, √(2) = 1.4, pi = 22/7, NA = 6 × 10^23 ]](https://toppr-doubts-media.s3.amazonaws.com/images/4700975/dc175a27-4ab9-44ce-88e1-7d9b75e43184.jpg)