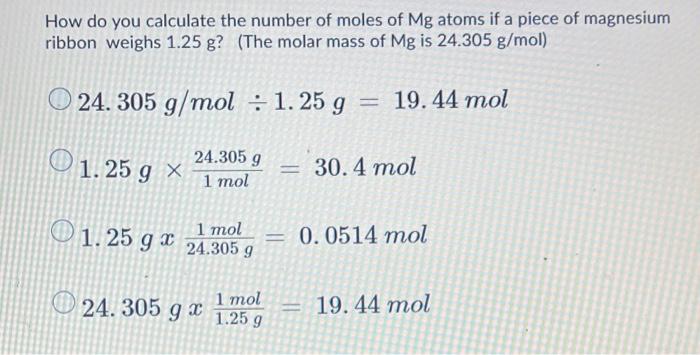Calculate the number of particles in each of the following: (a) 48 g of Mg (b) 8 g of O2 (c) 0.1 mole of carbon (Atomic mass Mg = 24 u, O =
![Whole-rock mg-number [(Mg/(Mg + Fe 2+ ) × 100] vs olivine composition... | Download Scientific Diagram Whole-rock mg-number [(Mg/(Mg + Fe 2+ ) × 100] vs olivine composition... | Download Scientific Diagram](https://www.researchgate.net/profile/Michael-Garcia-15/publication/237505959/figure/tbl3/AS:393280111038464@1470776693021/Microprobe-analyses-of-plagioclase-in-two-samples-from-vent-D-episode-54_Q320.jpg)
Whole-rock mg-number [(Mg/(Mg + Fe 2+ ) × 100] vs olivine composition... | Download Scientific Diagram
![Calculate the number of molecules present in 0.5 moles of magnesium oxide (MgO) . [Atomic weights : Mg = 24, O = 16 ] Calculate the number of molecules present in 0.5 moles of magnesium oxide (MgO) . [Atomic weights : Mg = 24, O = 16 ]](https://dwes9vv9u0550.cloudfront.net/images/2527137/b4ebf9c0-6b64-43b4-94e9-555f3180cc58.jpg)
Calculate the number of molecules present in 0.5 moles of magnesium oxide (MgO) . [Atomic weights : Mg = 24, O = 16 ]

Calculate the number of moles of magnesium present in a magnesium ribbon weighing 12 g. molar atomic mass of magnesium is 24 g mole^-1
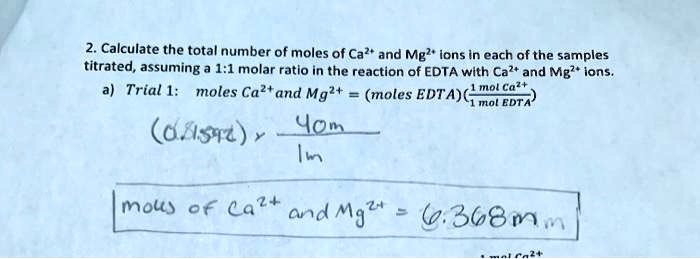
SOLVED: 2. Calculate the total number of moles of Ca" and Mg?' ions in each ofthe samples titrated, assuming 1;1 molar ratio in the reaction of EDTA with Cal+ and Mg?* ions

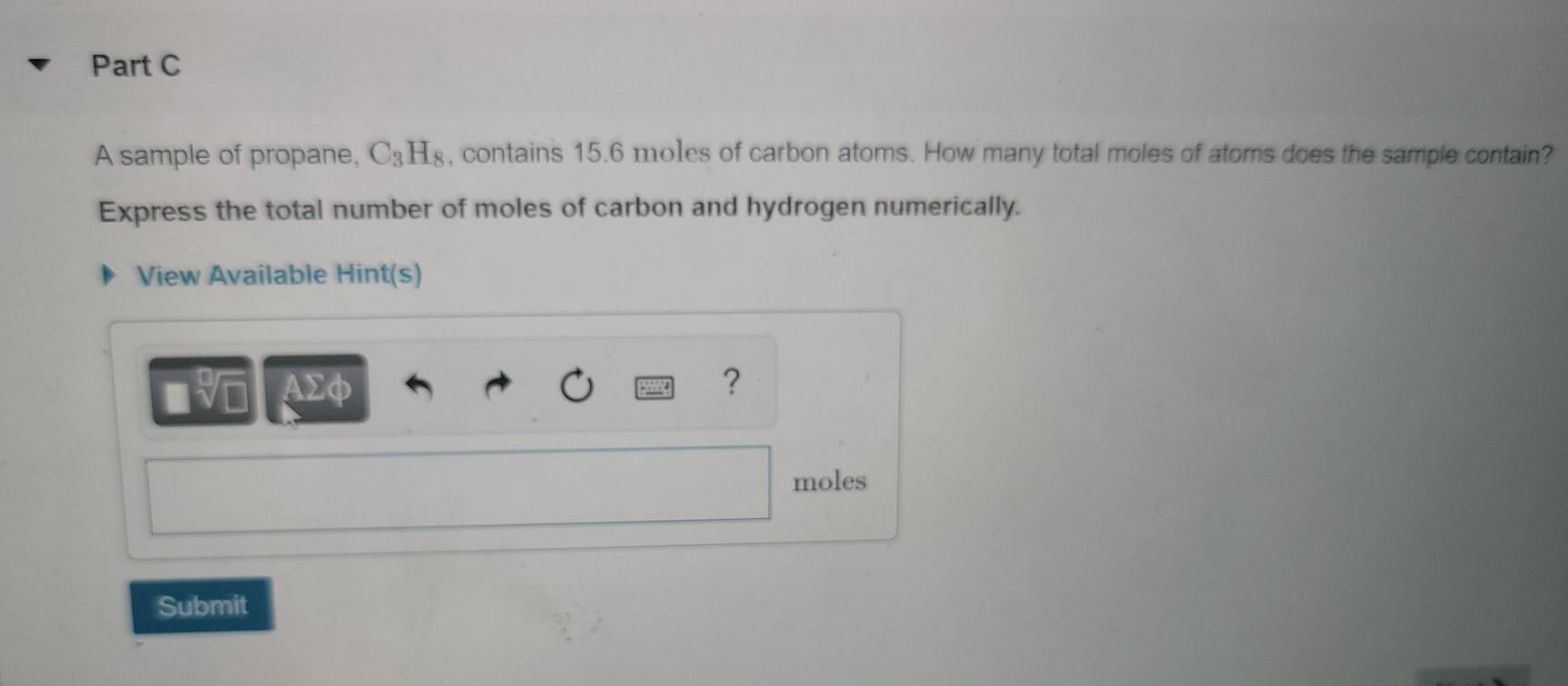
Calculate the number of moles of magnesium oxide, MgO in i. 80 g and ii. 10 g of the compound. (Average atomic masses of Mg = 24 and O = 16 )
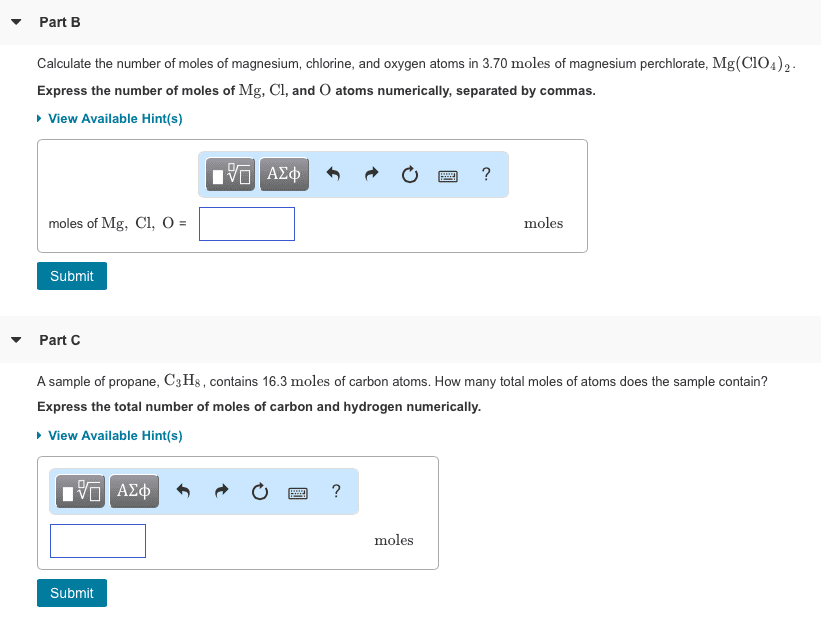
OneClass: Part B Calculate the number of moles of magnesium, chlorine, and oxygen atoms in 3.70 moles...
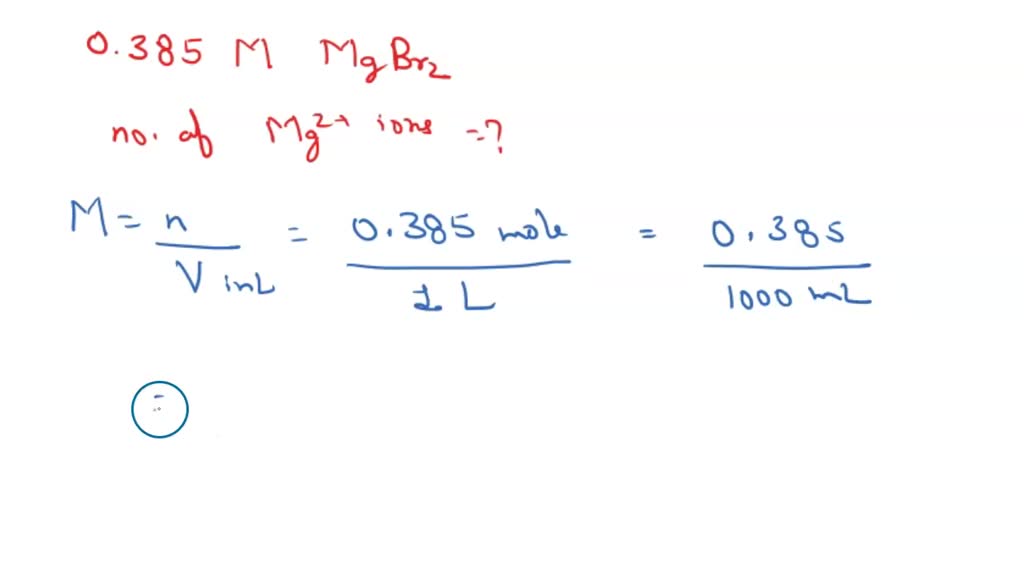
SOLVED: Calculate the following quantity: number of Mg2+ ions in each milliliter of 0.537 M magnesium bromide
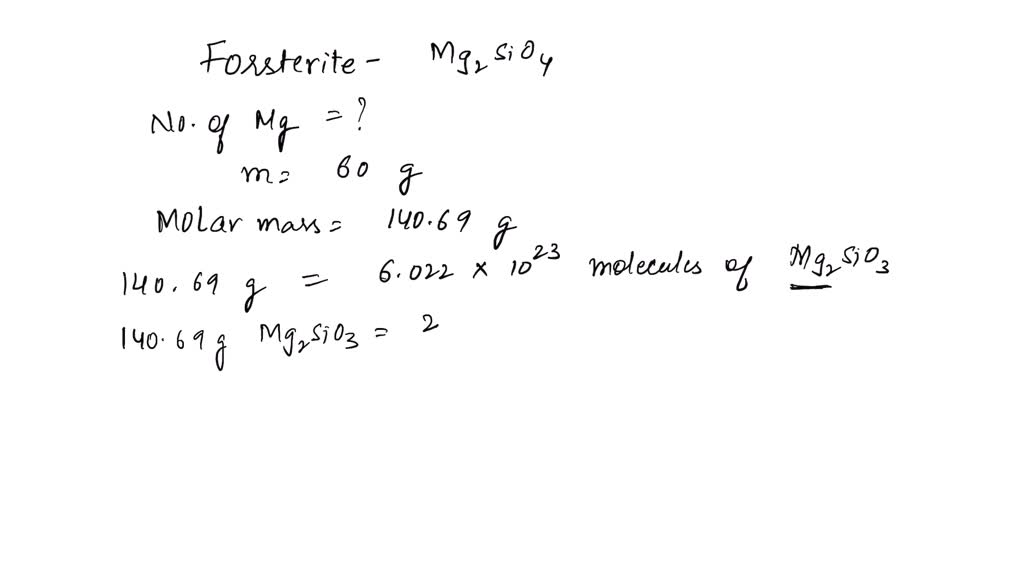
SOLVED: Calculate the number of magnesium atoms in a 60.0g sample of forsterite . Be sure your answer has a unit symbol if necessary, and round it to significant digits.

Calculate the mole ratio of `240 g` of calcium and `240 g` of magnesium. `(Ca = 40 , Mg = 24 u)` - YouTube