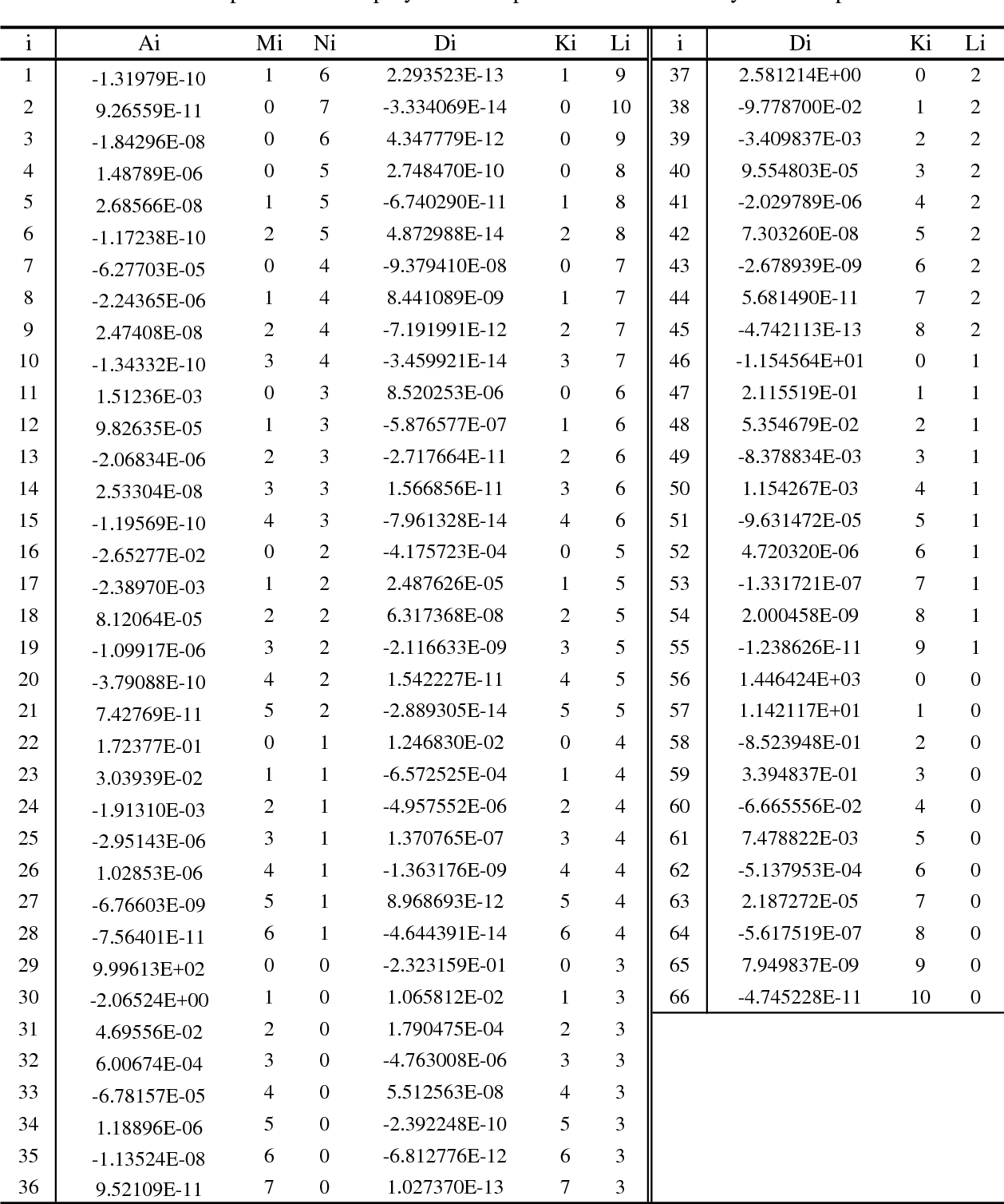
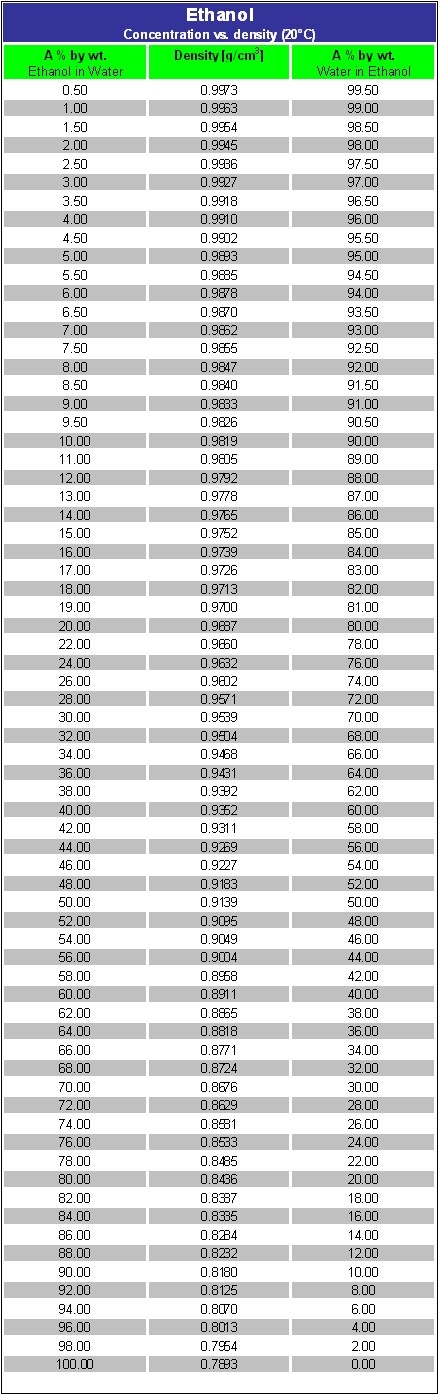
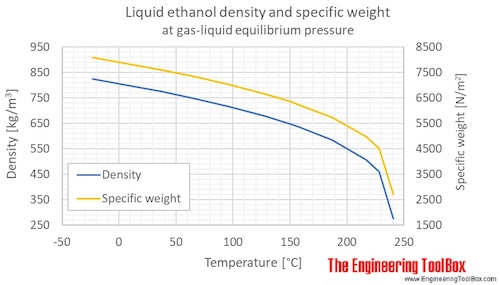
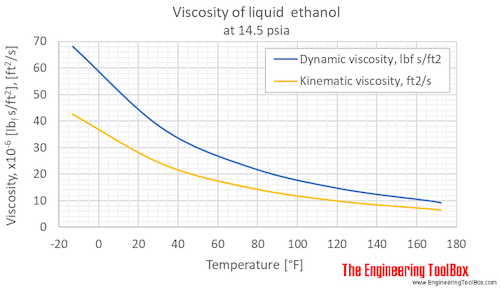
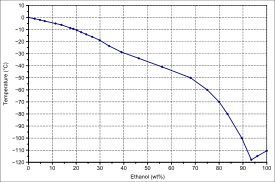
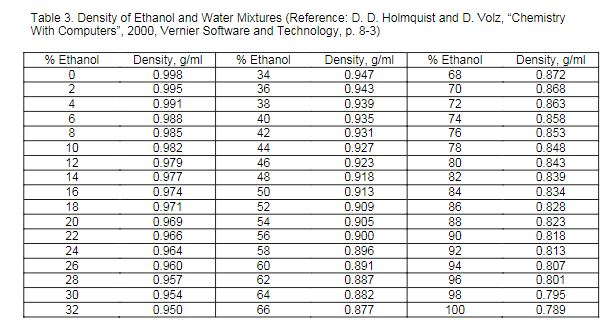
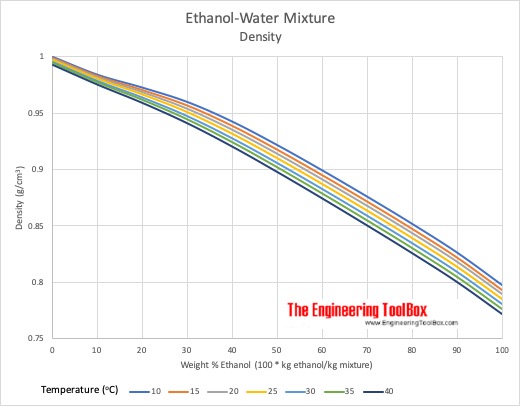
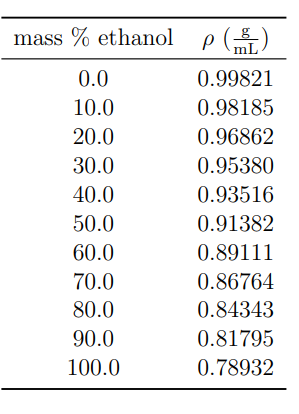
Computing the Composition of Ethanol-Water Mixtures Based on Experimental Density and Temperature Measurements
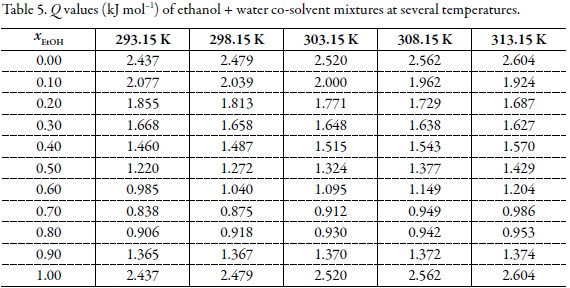
Preferential solvation of acetaminophen in ethanol + water solvent mixtures according to the inverse Kirkwood-Buff integrals method

The density of water and ethanol at room temperature is 1.0 g / mL and 0.789 g / mL respectively. What volume of ethanol contains the same number of molecules as are
Computing the Composition of Ethanol-Water Mixtures Based on Experimental Density and Temperature Measurements
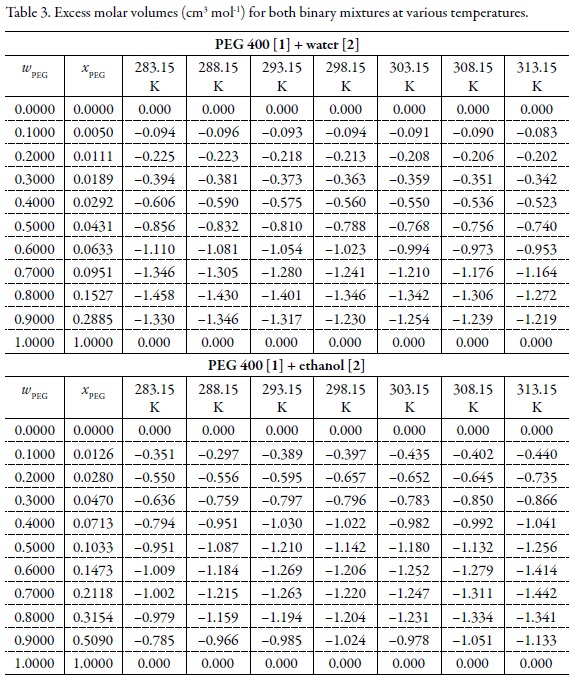
Volumetric properties of (PEG 400 + water) and (PEG 400 + ethanol) mixtures at several temperatures and correlation with the Jouyban-Acree model
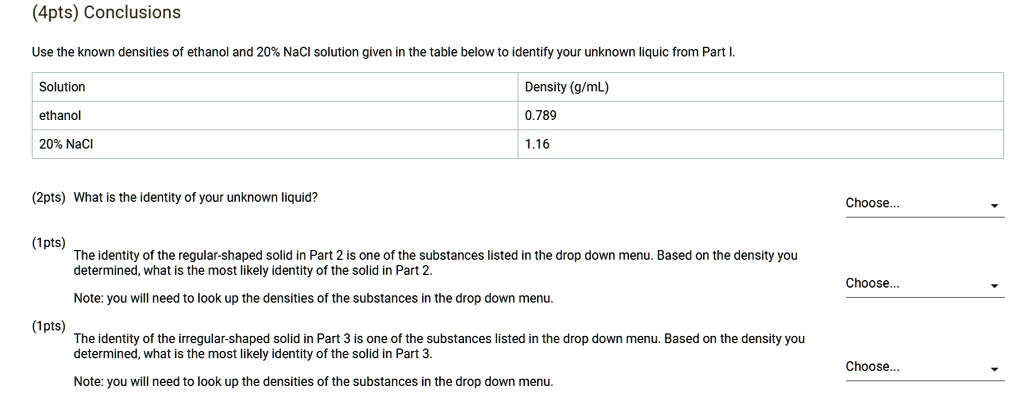
SOLVED: (Apts) Conclusions Use the known densities of ethanol and 20% NaCl solution given in the table below to identify your unknown Iiquic from Part !. Solution Density (g/mL) ethanol 0.789 20%