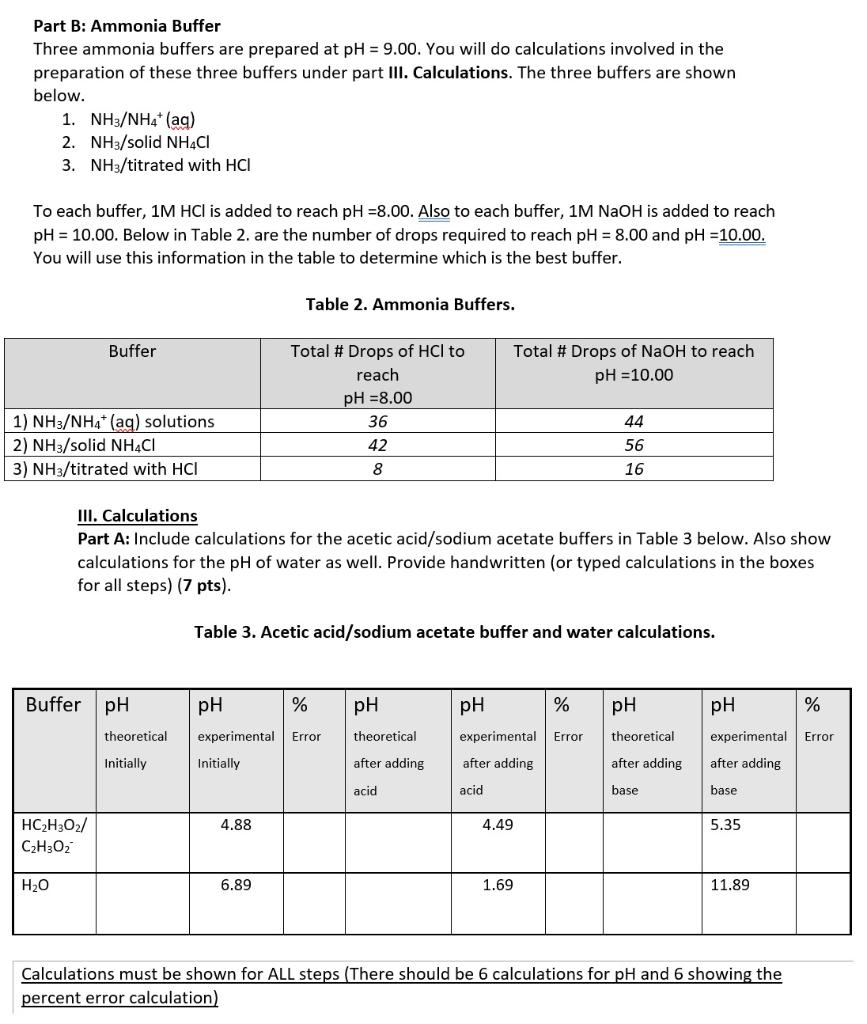
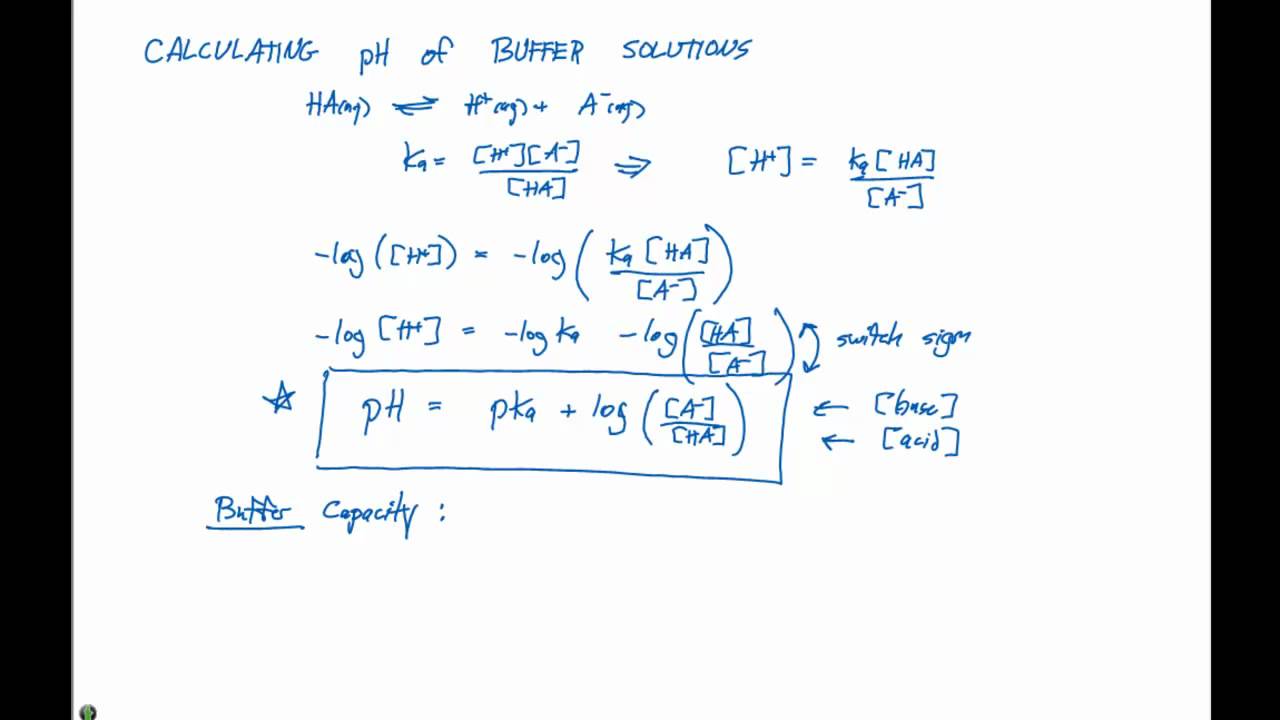
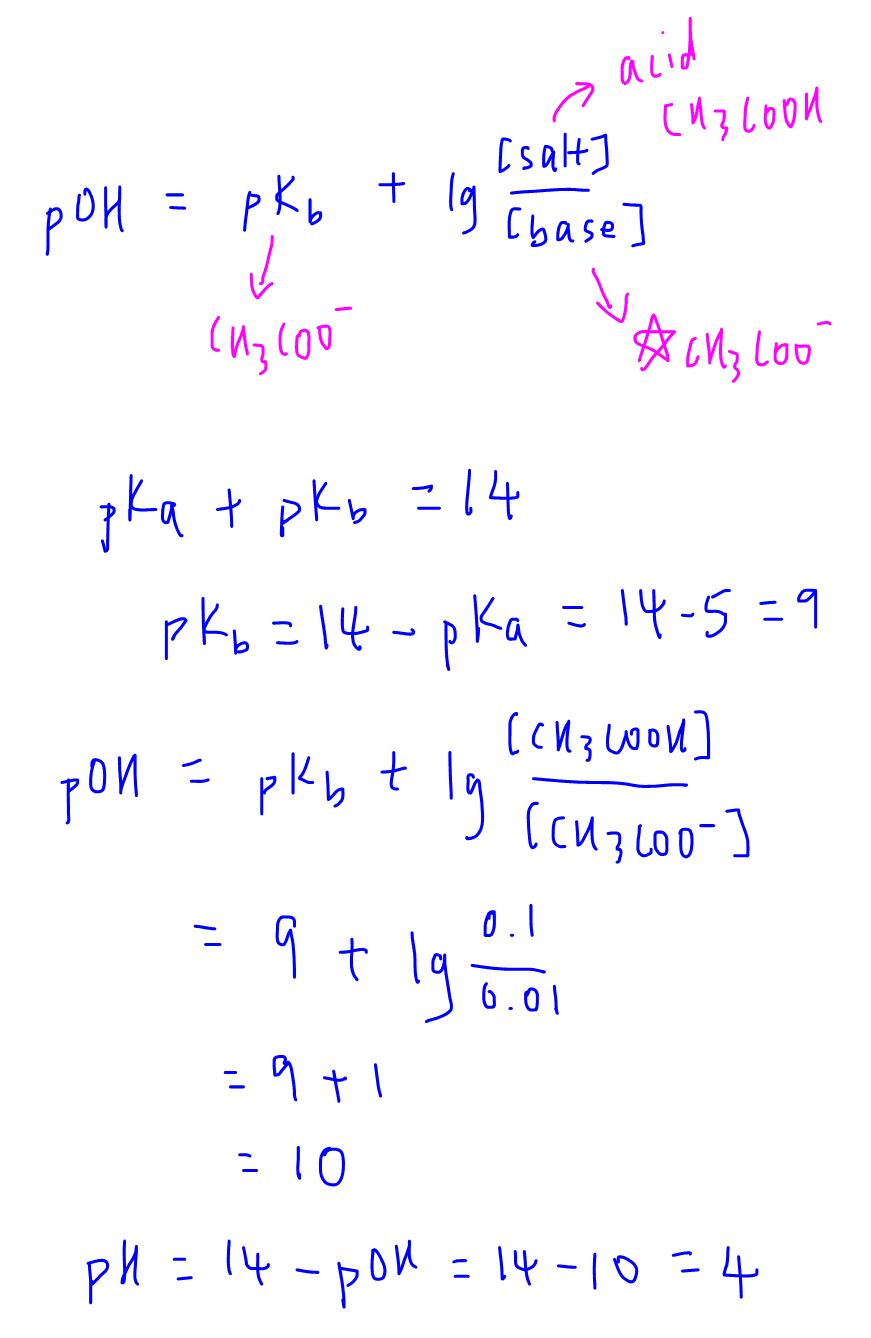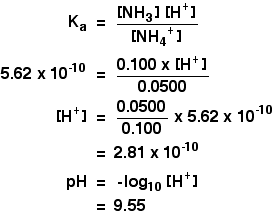SOLVED: Using the Henderson-Hasselbalch equation, calculate the predicted pH value for each buffer solution based on the volumes given in Tables 2, 3,and 4. The pKa of acetic acid is 4.75. Show

Acid-Base Buffers Equation & Examples | How to Calculate pH of a Buffer - Video & Lesson Transcript | Study.com